Summary
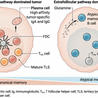
Binding of antigen to the B cell antigen receptor (BCR) initiates a multitude of events resulting in B cell activation. How the BCR becomes signaling-competent upon antigen binding is still a matter of controversy. Using a high-resolution proximity ligation assay (PLA) to monitor the conformation of the BCR and its interactions with co-receptors at a 10-20 nm resolution, we provide direct evidence for the opening of BCR dimers during B cell activation. We also show that upon binding Syk opens the receptor by an inside-out signaling mechanism that amplifies BCR signaling. Furthermore, we found that on resting B cells, the coreceptor CD19 is in close proximity with the IgD-BCR and on activated B cells with the IgM-BCR, indicating nanoscale reorganization of receptor clusters during B cell activation


 Your new post is loading...
Your new post is loading...




Talk at FEMS/EMBO Paris next week
http://www.febs-embo2014.org/programme/gene-diversification-and-immune-recognition/85
Sensing the unknown: How B cells are activated by a large set of different ligands
Since the discovery of antibodies (first in the form of antitoxins in 1890) immunologists have always wondered how our body can produce antibodies against so many different substances that they summarize with the word antigen. In 1956 Burnet proposed that a specific antibody response is established by a selection process that out of a large population of diverse B cells activates those with the right cognate B cell antigen receptor (BCR). The antigen-dependent stimulation of these cells results in their clonal outgrowth and differentiation to antibody producing plasma cells. How the BCR diversity is generated in the B cell population was explained by the finding of the V(D)J rearrangement and somatic hypermutation mechanisms of the immunoglobulin genes over the last decades. What is less well understood is how the many different antigens can activate the BCR in a way that is independent of the spatial organization of epitopes on the antigen molecules. This is quite different from most other receptors that have only one ligand whose structure helps to of bring or stabilize the receptor in an active conformation. As a solution for this problem, we have proposed the Dissociation Activation Model (DAM) according to which it is the opening of a pre-organized receptor cluster (oligomer) that activates the BCR independently of the structural input of the antigen. With the Fab base proximity ligation assay (Fab-PLA) we have developed a method which allows us to reliably monitor the conformation of the BCR on the surface of normal B cells at 10-20 nanometer distances. With this and other methods we found that the BCR exits indeed as an ordered oligomer on the surface of B cells and that in contact to antigen this oligomer is opened thus initiating the signaling process. Furthermore we discovered that the majority of the roughly 120.000 BCR on the surface of B cells are not opened by antigen but rather by the spleen tyrosine kinase (Syk) via an inside-out signaling mechanism. The details of this process will be discussed during my lecture.
Yang, J. and Reth, M. (2010). The dissociation activation model of B cell antigen receptor triggering. FEBS Letters 584: 4872. Klaesener, K., P. Maity, et al. (2014). "B cell activation involve nanoscale receptor reorganizations and i out signaling by Syk." eLife, on-line in June.